Crystal Angel Ornament Science Fair Project
One of the defining features of water is its solubility, meaning its ability to dissolve other substances.
This property plays a critical role in many chemical reactions and biological processes.
In this experiment, you will explore the solubility of Borax powder by dissolving it in water to create a saturated solution.
The term “saturated” means that no more powder can be absorbed by the water.
You will use hot water to increase the solubility of the powder and then let the solution cool down.
As the water cools, the Borax powder will begin to precipitate and solidify, turning from a liquid state to a solid form.
The precipitated powder will stick together and form beautiful crystals on the pipe cleaner and the sides of the glass.
These crystals can create stunning crystal angel ornaments.

Grow A Crystal Angel Ornament
This is a fun experiment for kids.
Materials
- Borax (found in laundry section)
- colorful pipe cleaners (dollar store)
- water
- string
- a pencil or a stick
Tools
- wide-mouth jar or glass
- imagination
- adult supervision
Instructions
How to make crystal angels
- Bend a pipe cleaner into the shape you want to make a crystal out of. This is the step you can use your imagination.

- Fill the glass with water to around three quarters full. Then use a measuring cup to measure the amount of water used.
- Boil the water in a pan under adult supervision
- Carefully pour the boiling water into the glass.
- Dissolve plenty of Borax into the hot water. For each cup of water, add 3 tablespoons of Borax.
- Stir the water until all the powder has dissolved or the powder left at the bottom cannot dissolve further. This phenomenon is called saturation.
- Tie the shape you previously made with a string and hang the shape at the mouth of the glass using the pencil or the stick. Make sure the pipe cleaner is fully submerged and not touching the side of the glass.
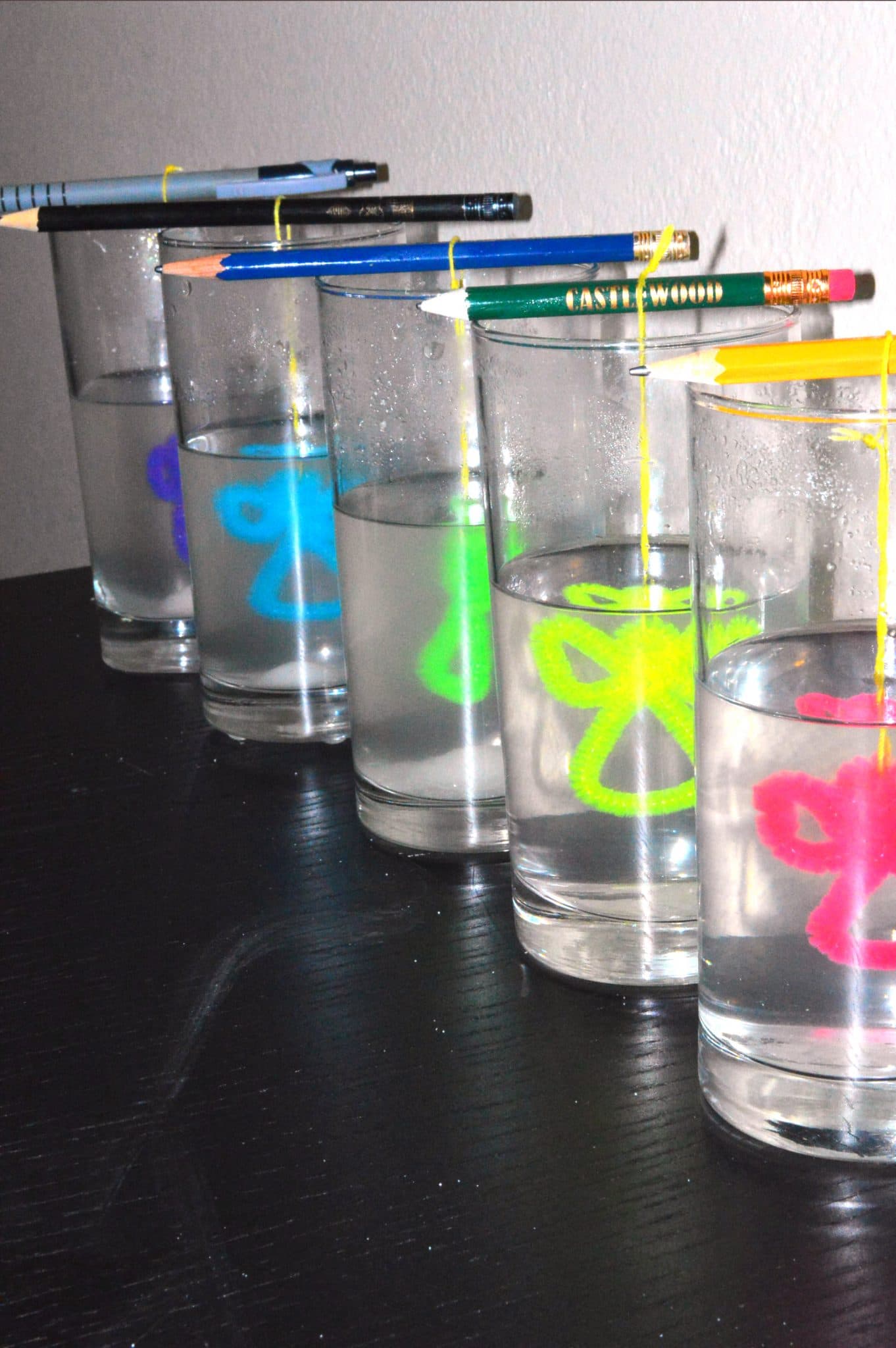
- Put the glass at an area where it won't be disturbed for the next hour.
- Patiently wait.

Notes
Unlike growing crystals using sugar or salt, crystallization using Borax is relatively fast. Crystals can be seen forming as early as two hours after the glass is set aside.
Monitor the progress from time to time to get the desired thickness of crystals. If left overnight, it may become too thick.
Recommended Products
As an Amazon Associate, I earn from qualifying purchases.
By repeating the process multiple times, you can make the crystals grow bigger and create a more intricate design.
You can experiment with different temperatures, solvents, and solutes to observe the effect on the crystal formation.
This simple experiment can help you understand the concept of saturation, solubility, and precipitation, which are important principles in chemistry.
For more scientific explanations on crystallization, see Nucleation and Crystal Growth. Find more science experiments with water here: Best Science Experiments With Water









